Description
Perfect Starter Pen
✓ In Stock
48hr UK Dispatch
COA Included
Cold-Chain Delivery
Research Use Only
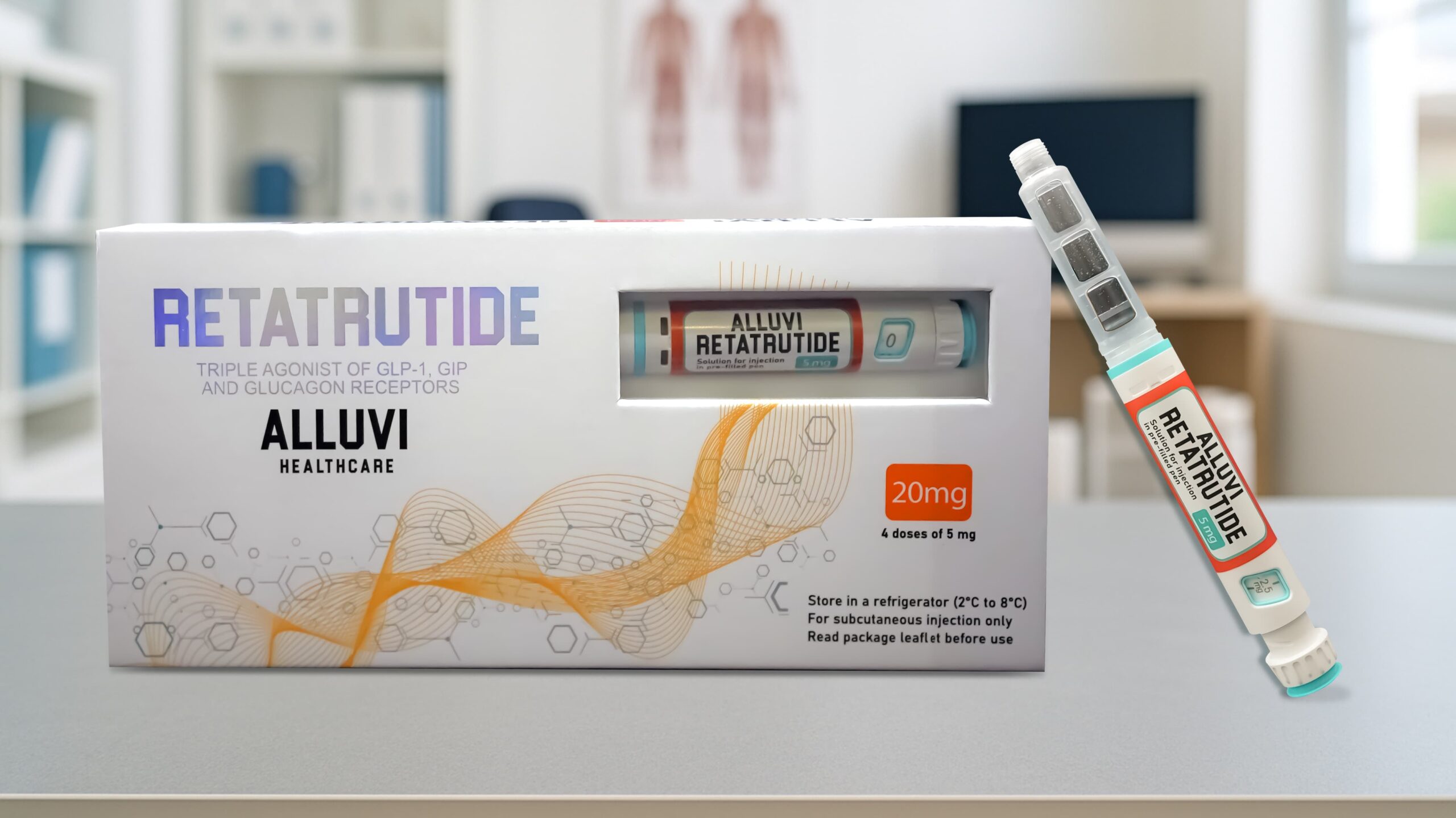
The Alluvi Retatrutide 20mg Pen is the ideal entry-point retatrutide pen UK for researchers beginning a structured retatrutide protocol — a pre-filled, Synedica-manufactured, research-grade subcutaneous injection pen containing 20mg of triple GLP-1/GIP/Glucagon receptor agonist retatrutide. Independently verified at ≥98% purity with a batch-specific Certificate of Analysis included in every order, the Alluvi Retatrutide 20mg Pen is supplied exclusively by Alluvi Healthcare — the UK’s most trusted retatrutide UK source. Dispatched within 48 hours from UK stock with cold-chain tracked delivery. For UK researchers asking where to buy retatrutide UK at the initiation phase — this is the answer. For research and educational purposes only. Not for human or veterinary consumption.
What Is the Alluvi Retatrutide 20mg Pen?
The Alluvi Retatrutide 20mg Pen is a pre-filled, Synedica-manufactured, research-grade subcutaneous injection pen containing 20mg of retatrutide (LY3437943) — the world’s most studied triple GLP-1/GIP/Glucagon receptor agonist peptide. Supplied exclusively in the United Kingdom by Alluvi Healthcare, the Alluvi Retatrutide 20mg Pen is the go-to initiation-phase reta pen UK for researchers entering the retatrutide compound range for the first time, or for those conducting shorter-duration or pilot research protocols.
As the companion product to the Alluvi Retatrutide 40mg Pen, the 20mg pen delivers the same Synedica-quality retatrutide in a smaller format — ideal for the first 4–8 weeks of a structured escalation protocol before transitioning to the higher-capacity retatrutide 40mg pen. Every Alluvi Retatrutide 20mg Pen is independently purity-verified, cold-chain dispatched from UK stock within 48 hours, and arrives complete with a batch-specific Certificate of Analysis.
Whether you are buying retatrutide for the first time, running a pilot study, or managing a multi-phase research programme, the Alluvi Retatrutide 20mg Pen from Alluvi Healthcare gives you the most reliable, best-documented, and most cost-efficient entry into the retatrutide pens UK market. The UK’s trusted retatrutide UK source — buyalluvipeptides.co.uk.
What’s Included — Alluvi Retatrutide 20mg Pen Research Kit
Every Alluvi Retatrutide 20mg Pen is dispatched as a complete research kit from UK stock in discreet, cold-chain insulated, unmarked outer packaging. Everything needed to begin a retatrutide initiation protocol is included:
- 1 × Pre-filled Alluvi Retatrutide 20mg Pen — Synedica manufactured, precision click-dial mechanism, subcutaneous injection format
- 8 × Sterile single-use disposable pen needles — compatible with the Alluvi pen format
- 1 × Batch-specific Certificate of Analysis (COA) — independent third-party purity verification confirming ≥98% retatrutide
- 1 × Alluvi Research Information Sheet — compound overview, initiation-phase protocol reference, storage and handling guidance
- Cold-pack insulated thermal liner — maintains 2–8°C throughout UK transit
- Discreet unmarked outer packaging — no brand name, no product indicators on the outer box
- Full tracked delivery label — Royal Mail / DPD tracking from dispatch to your door
Full Technical Specifications — Alluvi Retatrutide 20mg Pen
The following specifications apply to every Alluvi Retatrutide 20mg Pen supplied by Alluvi Healthcare. All Synedica retatrutide pen units are manufactured under controlled pharmaceutical conditions and independently verified before UK supply.
| Product Name | Alluvi Retatrutide 20mg Pen |
| Manufacturer | Synedica (distributed by Alluvi Healthcare UK) |
| Active Compound | Retatrutide (LY3437943) |
| Compound Class | Triple GLP-1 / GIP / Glucagon Receptor Agonist |
| Total Retatrutide Per Pen | 20mg |
| Dosage Increments | 2.5mg · 5mg · 7.5mg · 10mg (precision click-dial) |
| Maximum Full Doses Per Pen | 2 × 10mg doses |
| Maximum Micro-Doses Per Pen | Up to 8 × 2.5mg increments |
| Delivery Format | Pre-filled subcutaneous injection pen |
| Needle Type | Fine-gauge subcutaneous pen needle (8 included) |
| Purity Standard | ≥98% (independent third-party lab verification) |
| Certificate of Analysis | Batch-specific COA included with every order |
| Storage — Unopened | Refrigerated 2–8°C, away from direct light. Do not freeze. |
| Storage — In Use | Room temperature <30°C for up to 28 days, or return to refrigeration |
| Shelf Life | 24 months from date of manufacture (see pen label) |
| Ideal Protocol Phase | Initiation Phase (Weeks 1–8) · Pilot & Short-Duration Studies |
| Weeks Supply (at 2.5mg/wk) | 8 weeks |
| Weeks Supply (at 5mg/wk) | 4 weeks |
| Research Use Only | Not for human or veterinary consumption |
| UK Dispatch Time | Within 48 hours of order confirmation |
| UK Delivery | 1–2 working days from dispatch, fully tracked |
| Packaging | Cold-chain insulated, discreet unmarked outer box |
| Supplier | Alluvi Healthcare — buyalluvipeptides.co.uk |
The Science — What Is Retatrutide & Why Does the 20mg Pen Matter?
Retatrutide (LY3437943) is a synthetic acylated peptide and the world’s most advanced triple receptor agonist, simultaneously activating GLP-1, GIP, and Glucagon hormone pathways in a single molecule. The Alluvi Retatrutide 20mg Pen delivers this compound in a starter-format pen designed specifically for the critical initiation phase of any retatrutide research protocol — the phase that determines compound tolerance, baseline receptor response data, and optimal dose escalation timing for subsequent phases.
Understanding the science behind retatrutide is essential for any UK researcher choosing between the Alluvi Retatrutide 20mg Pen and the Alluvi Retatrutide 40mg Pen — or evaluating retatrutide against Synedica Tirzepatide for their specific research programme.
Triple Receptor Agonism — The Mechanism Behind the 20mg Pen
1. GLP-1 Receptor Agonism
Glucagon-like peptide-1 (GLP-1) receptor activation produces potent appetite signal suppression in the hypothalamus, slowing of gastric emptying, enhancement of glucose-stimulated insulin secretion, and improvement of overall glycaemic control. The GLP-1 agonism in the Alluvi Retatrutide 20mg Pen is structurally optimised to deliver significantly greater receptor engagement than first-generation GLP-1 research compounds. At the 2.5mg and 5mg doses delivered by the 20mg pen during the initiation phase, GLP-1 receptor engagement is already measurably more potent than equivalent semaglutide doses in published comparative studies.
2. GIP Receptor Agonism
Glucose-dependent insulinotropic polypeptide (GIP) receptor agonism — the mechanism that distinguishes tirzepatide from semaglutide — is built into retatrutide from the ground up. GIP agonism supports insulin release, enhances fat metabolism in combination with GLP-1 signalling, and improves insulin sensitivity in peripheral tissues. Even at the initiation-phase doses delivered by the Alluvi Retatrutide 20mg Pen, GIP receptor engagement begins contributing to the compound’s unique dual metabolic signalling profile from week one of the research protocol.
3. Glucagon Receptor Agonism
The third and most distinctive pathway — absent from Synedica Tirzepatide and all other UK-available dual-agonist research compounds — is glucagon receptor activation. Even at the lower initiation doses in the Alluvi Retatrutide 20mg Pen, glucagon receptor engagement begins elevating resting energy expenditure, promoting independent lipolysis, and raising basal metabolic rate. This early glucagon receptor data collected during the 20mg pen phase becomes foundational baseline data for subsequent high-dose research phases conducted with the Alluvi Retatrutide 40mg Pen.
Alluvi Retatrutide 20mg Pen — The Ideal Initiation Phase Research Tool
The Alluvi Retatrutide 20mg Pen is specifically designed for the first 4–8 weeks of a structured retatrutide research programme — the initiation phase. This phase is the most critical part of any retatrutide protocol: it establishes compound baseline data, identifies individual receptor response profiles, and documents tolerance progression before escalation to higher-dose phases.
Here is why the Alluvi Retatrutide 20mg Pen is the optimal UK research tool for the initiation phase:
- Perfect dose range for initiation: The 20mg pen delivers 8 × 2.5mg doses or 4 × 5mg doses — precisely the dose range documented in published retatrutide initiation protocols (2.5mg weeks 1–4, 5mg weeks 5–8).
- Cost-efficient for pilot studies: Researchers running pilot or feasibility studies with smaller subject cohorts benefit from the 20mg pen’s lower per-pen cost relative to the 40mg format when full-dose capacity is not required.
- Ideal for comparative research: The 20mg pen’s dose range perfectly overlaps with the initiation and first titration phases of Synedica Tirzepatide protocols, making it ideal for comparative dual-vs-triple agonist initiation-phase studies.
- Foundation for 40mg pen transition: All retatrutide data generated using the Alluvi Retatrutide 20mg Pen in weeks 1–8 becomes the baseline for transitioning to the Alluvi Retatrutide 40mg Pen in the titration and maintenance phases.
- Flexible micro-dosing: The click-dial mechanism enables precise 2.5mg increment dosing, essential for careful receptor response mapping in the early stages of novel peptide research.
- Same Synedica quality as the 40mg pen: The Alluvi Retatrutide 20mg Pen is manufactured by the same Synedica facility to the same quality standards as the 40mg pen — ≥98% purity, batch COA, cold-chain supply chain.
Retatrutide Research Dosage Protocol — 20mg Pen Phase (Scientific Reference)
The following dosage schedule reflects the staged escalation protocols documented in peer-reviewed retatrutide research. This is provided as scientific and educational reference only — not medical advice. The Alluvi Retatrutide 20mg Pen is for laboratory research use only and is not for human consumption.
| Research Phase | Weeks | Reference Dose | Doses From 20mg Pen | Research Notes |
|---|---|---|---|---|
| Phase 1 — Initiation | 1–4 | 2.5mg | 8 doses (full 4-week supply) | Baseline data collection; initial receptor response mapping |
| Phase 2 — Titration I | 5–8 | 5mg | 4 doses (full 4-week supply) | First escalation step; GIP & GLP-1 response comparison |
| Phase 3 — Titration II * | 9–12 | 7.5mg | 2–3 doses (partial) | * Transition to 40mg pen recommended from this phase onwards |
| Phase 4 — Maintenance * | 13–48+ | 10mg | 2 doses | * Use Alluvi Retatrutide 40mg Pen for cost-efficiency at this phase |
The Alluvi Retatrutide 20mg Pen is optimally used for Phases 1 and 2 of the escalation protocol — covering the complete 8-week initiation and first titration period. From Phase 3 onwards, researchers typically transition to the Alluvi Retatrutide 40mg Pen for cost-efficient, higher-capacity dosing across the maintenance phase.
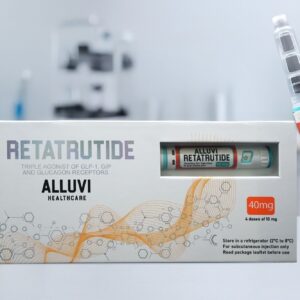
Weeks 9 onwards: Transition to the Alluvi Retatrutide 40mg Pen for Titration II (7.5mg) and Maintenance (10mg) phases. This two-pen structured approach is consistent with published escalation methodology and maximises research cost-efficiency. View the Alluvi Retatrutide 40mg Pen →
The Complete Retatrutide Research Journey — From 20mg to 40mg Pen
Understanding how the Alluvi Retatrutide 20mg Pen fits into the broader retatrutide research journey is essential for UK researchers planning multi-phase protocols. The journey from initiation to maintenance using Alluvi Healthcare’s full retatrutide pens UK range follows this structured progression:
How to Use the Alluvi Retatrutide 20mg Pen — Step-by-Step Research Protocol
The Alluvi Retatrutide 20mg Pen is pre-filled and ready to use immediately — no reconstitution, no mixing, no preparation required beyond standard laboratory hygiene protocols. Its precision click-dial mechanism ensures accurate, repeatable dosing for high-integrity initiation-phase research.
- Remove from refrigeration: Take the Alluvi Retatrutide 20mg Pen from the refrigerator 15–30 minutes before use. Allow it to reach ambient room temperature naturally — do not heat artificially. This reduces viscosity and ensures consistent compound flow during injection.
- Visual inspection: Examine the pen through its clear window. The retatrutide solution should appear clear to slightly opalescent and completely free of visible particles or discolouration. If the solution appears cloudy, discoloured, or if the pen has been frozen at any point, do not use — contact Alluvi Healthcare on +44 7537 823566.
- Attach a sterile needle: Select a fresh sterile needle from the 8-pack supplied with your kit. Remove the outer protective seal. Screw firmly clockwise onto the pen tip until fully secure. Never reuse needles between research sessions.
- Prime the pen: Select a small prime amount using the dose dial. Hold the pen upright with the needle pointing upward and press the injection button to expel trapped air. A small drop of solution at the needle tip confirms successful priming and a clear flow pathway.
- Set your research dose: Rotate the click-dial to the required research dose — 2.5mg for initiation phase, 5mg for first titration phase. Confirm the dose is clearly displayed in the dose window before proceeding.
- Select the injection site: Choose an appropriate subcutaneous site — the periumbilical abdomen (minimum 5cm from the navel), the anterolateral thigh, or the posterior upper arm. Rotate sites systematically between research sessions to minimise localised tissue response. Wipe the selected site with an alcohol swab and allow to dry fully.
- Administer: Hold the pen at a 90-degree angle to the skin surface. Insert the needle with a smooth, controlled motion. Press the injection button fully and hold for a full 5–10 second count before withdrawing — this ensures the complete dose is delivered. Apply gentle pressure to the site with a clean swab if needed.
- Post-injection protocol: Unscrew and dispose of the used needle immediately in a certified sharps container. Replace the pen cap securely. Record the dose, date, and injection site in your research log. Return the Alluvi Retatrutide 20mg Pen to refrigerated storage at 2–8°C.
Storage & Handling — Alluvi Retatrutide 20mg Pen
Correct storage of the Alluvi Retatrutide 20mg Pen is critical to maintaining compound purity and research validity. The retatrutide peptide is sensitive to temperature extremes and light exposure. Alluvi Healthcare cold-chain dispatches every pen order, but researcher storage responsibility begins the moment the package is received.
- Unopened storage: Refrigerate immediately upon receipt at 2–8°C. Store upright in original packaging, protected from direct light. Shelf life up to 24 months from manufacture date (printed on pen label).
- In-use storage: An opened Alluvi Retatrutide 20mg Pen may be kept at room temperature (maximum 30°C) for up to 28 days in controlled research conditions. Mark the first use date on the pen barrel. Return to refrigeration between sessions where possible.
- Never freeze: Freezing will permanently and irreversibly denature the retatrutide peptide structure. A frozen pen cannot be recovered — it must be discarded. If you suspect freezing during transit, do not use and contact Alluvi Healthcare immediately.
- Light protection: UV exposure and prolonged fluorescent laboratory lighting will degrade the retatrutide compound over time. Always store with the pen cap secured and keep away from direct sunlight, windows, and UV-emitting laboratory equipment.
- Transport: When transporting between research locations, use a qualified pharmaceutical insulated carrier with a cold pack to maintain 2–8°C. Do not transport the pen without cold-chain protection.
- Disposal: Expired or depleted Alluvi Retatrutide 20mg Pens should be disposed via your institution’s chemical/pharmaceutical research waste contractor in compliance with applicable UK waste regulations.
Alluvi Retatrutide 20mg Pen vs 40mg Pen vs Synedica Tirzepatide — Full Comparison
For UK researchers comparing available research compounds from Alluvi Healthcare, the following table provides a direct comparison between the Alluvi Retatrutide 20mg Pen, the Alluvi Retatrutide 40mg Pen, and Synedica Tirzepatide.
| Feature | Alluvi Retatrutide 20mg Pen | Alluvi Retatrutide 40mg Pen | Synedica Tirzepatide |
|---|---|---|---|
| GLP-1 Agonism | ✓ Yes | ✓ Yes | ✓ Yes |
| GIP Agonism | ✓ Yes | ✓ Yes | ✓ Yes |
| Glucagon Agonism | ✓ Yes | ✓ Yes | ✗ No |
| Total Retatrutide | 20mg | 40mg | N/A |
| Full 10mg Doses | 2 doses | 4 doses | N/A |
| Ideal Protocol Phase | Initiation · Pilot Studies | Advanced · Maintenance | Intermediate · Comparison |
| Weeks Supply (2.5mg/wk) | 8 weeks | 16 weeks | Varies |
| COA Included | ✓ Every order | ✓ Every order | ✓ Every order |
| UK Stock at Alluvi | ✓ In Stock | ✓ In Stock | ✓ In Stock |
| Best For | New researchers, starter protocols | Experienced researchers, extended studies | Dual-agonist comparative studies |
| Buy From | buyalluvipeptides.co.uk | buyalluvipeptides.co.uk | buyalluvipeptides.co.uk |
Why Buy the Alluvi Retatrutide 20mg Pen from Alluvi Healthcare — The UK’s #1 Retatrutide Source
When UK researchers look to buy retatrutide UK — whether for a first-time initiation study or an ongoing multi-phase programme — the choice of supplier matters enormously. Here is why Alluvi Healthcare is the definitive retatrutide UK source for the Alluvi Retatrutide 20mg Pen and the entire retatrutide range.
- Synedica-Certified Quality: Every Alluvi Retatrutide 20mg Pen is manufactured by Synedica — the same manufacturer behind the Synedica Retatrutide 40mg Pen and Synedica Tirzepatide. Batch-specific COA confirming ≥98% purity included with every order, automatically — no request required.
- 100% UK-Based Inventory: Alluvi Healthcare holds genuine UK stock of the retatrutide 20mg UK pen. No overseas imports, no customs complications, no cold-chain breaches from international transit. When you buy retatrutide UK from Alluvi, the pen is already in the UK before you order it.
- Guaranteed 48-Hour UK Dispatch: Every Alluvi Retatrutide 20mg Pen order is dispatched within 48 hours of order confirmation via tracked, cold-chain delivery. Most UK orders arrive within 1–2 working days of dispatch.
- Expert UK Research Support: Our research support team understands the retatrutide compound, initiation-phase protocols, and the full Synedica range. Available Monday to Saturday on +44 7537 823566.
- Competitive Pricing: Transparent pricing on the retatrutide 20mg UK pen — among the most competitive in the UK market, with no hidden charges. Free tracked UK delivery on orders over £150.
- Complete Discretion: Every retatrutide pens UK order dispatched in plain, unmarked outer packaging — no brand identification, no product indication visible on the exterior.
- Full Research Ecosystem: Alluvi Healthcare stocks everything you need for a complete retatrutide research programme: Alluvi Retatrutide 40mg Pen, Synedica Tirzepatide, Alluvi Supplement Box, Gummy Range, and the Alluvi Research App.
- Wholesale & Bulk Research Pricing: Research institutions, laboratories, and high-volume buyers can access competitive bulk pricing on all retatrutide pens UK. Contact us via buyalluvipeptides.co.uk/contact or call +44 7537 823566.
Where to Buy Retatrutide 20mg UK — Buyer’s Guide for First-Time Researchers
For researchers searching where to buy retatrutide UK for the first time — or specifically looking for the retatrutide 20mg UK format — the following guidance covers everything you need to know before making your first retatrutide buy UK.
What to Look for in a Retatrutide 20mg UK Source
- Confirmed manufacturer identity: The Alluvi Retatrutide 20mg Pen is manufactured by Synedica — a named, verifiable pharmaceutical manufacturer. Demand manufacturer transparency from any retatrutide UK source before purchasing.
- Batch-specific COA — not generic: A credible supplier of retatrutide pens UK provides a batch-specific Certificate of Analysis linking purity test results to the exact batch in your pen. Alluvi Healthcare does this automatically with every order.
- UK-based stock: Genuine UK inventory means 48-hour dispatch, no customs risk, and no cold-chain temperature breach from international transit. Critical for a temperature-sensitive compound like retatrutide.
- Cold-chain dispatch: Any retatrutide UK buy should arrive in cold-chain insulated packaging. Retatrutide begins to degrade above 8°C. Alluvi Healthcare cold-chain dispatches every single order as standard.
- Research-only compliance: Any UK supplier of retatrutide 20mg UK products must clearly position their products as research compounds only. Suppliers making medical or therapeutic claims are operating outside UK regulatory guidelines.
- Responsive UK support: Alluvi Healthcare’s research support team is reachable on +44 7537 823566 Monday to Saturday — direct, knowledgeable, UK-based.
The Full Alluvi Healthcare Research Range — Beyond the 20mg Pen
The Alluvi Retatrutide 20mg Pen is the ideal starting point, but Alluvi Healthcare’s full research range gives UK researchers everything they need to design, execute, and scale comprehensive retatrutide and metabolic peptide studies. All products below are UK-stocked and available from buyalluvipeptides.co.uk.
💉
Verified UK Researcher Reviews — Alluvi Retatrutide 20mg Pen
The following reviews are from verified UK researchers who have purchased the Alluvi Retatrutide 20mg Pen from Alluvi Healthcare. All reviews relate to product quality, delivery, and research experience — not medical outcomes.
“Started my retatrutide protocol with the Alluvi 20mg pen — perfect for the initiation phase. Arrived cold, COA bang on, and the research sheet included was genuinely useful for structuring weeks 1–8. Now onto the 40mg pen and couldn’t be happier.”
“Best place to buy retatrutide 20mg UK. Ordered Tuesday, arrived Wednesday in perfect condition — cold pack still frozen. COA confirmed ≥98% purity. Alluvi Healthcare is the only UK retatrutide source I trust.”
“Used the 20mg pen for a pilot comparative study against Synedica Tirzepatide. The retatrutide pen quality is exceptional — identical batch-to-batch. Alluvi Healthcare is our lab’s permanent UK retatrutide source for both 20mg and 40mg pens.”
“The Alluvi Retatrutide 20mg pen is exactly what I needed for the initiation phase of my 48-week protocol. Clean click mechanism, clear dose window, and arrived in discreet packaging with everything included. Seamless experience start to finish.”
“Switched to Alluvi after a bad experience with a no-name retatrutide UK source — no COA, no cold chain, questionable purity. Alluvi Healthcare is completely different league. The 20mg pen is the real deal.”
“Ordered 3 × Alluvi Retatrutide 20mg UK pens for a multi-subject initiation-phase study. All arrived next day, all cold, all with batch-specific COAs. The support team even helped me structure the protocol over the phone. Outstanding service.”
Research References — Scientific & Regulatory Context for the Alluvi Retatrutide 20mg Pen
The following authoritative external resources provide the scientific and regulatory context relevant to the Alluvi Retatrutide 20mg Pen and retatrutide research in the United Kingdom. All links are provided for educational reference purposes only.
🔬
Frequently Asked Questions — Alluvi Retatrutide 20mg Pen
The Alluvi Retatrutide 20mg Pen is a pre-filled, Synedica-manufactured, research-grade subcutaneous injection pen containing 20mg of retatrutide (LY3437943) — a triple GLP-1/GIP/Glucagon receptor agonist peptide. Supplied exclusively by Alluvi Healthcare in the UK for laboratory research and development purposes only. Not for human or veterinary consumption.
You can buy retatrutide 20mg UK — specifically the Alluvi Retatrutide 20mg Pen — directly from Alluvi Healthcare at buyalluvipeptides.co.uk. UK-based stock, 48-hour dispatch, cold-chain tracked delivery, COA included. Call our research support team: +44 7537 823566.
The Alluvi Retatrutide 20mg Pen contains 20mg of retatrutide and delivers 2 × full 10mg doses (or 8 × 2.5mg micro-doses). It is designed for initiation-phase protocols (weeks 1–8). The Alluvi Retatrutide 40mg Pen contains 40mg and delivers 4 × 10mg doses, designed for advanced titration and maintenance phases (weeks 9 onwards). Most researchers use both pens sequentially in a structured two-pen escalation programme.
The Alluvi Retatrutide 20mg Pen provides an 8-week supply at the initiation dose of 2.5mg per week (8 × 2.5mg doses), or a 4-week supply at the first titration dose of 5mg per week (4 × 5mg doses). This makes it a complete supply for the entire 8-week initiation and first titration phase documented in published retatrutide research.
All Alluvi Retatrutide 20mg Pen orders are dispatched from UK stock within 48 hours of order confirmation, via tracked cold-chain delivery (Royal Mail or DPD). Most UK orders arrive within 1–2 working days from dispatch. Full tracking number provided by email upon dispatch.
Yes. The Alluvi Retatrutide 20mg Pen is manufactured by Synedica and distributed in the UK exclusively by Alluvi Healthcare. It is made to the same Synedica quality standards as the Synedica Retatrutide 40mg Pen and Synedica Tirzepatide — independently purity-verified at ≥98% with a batch COA included.
For any new retatrutide research programme, Alluvi Healthcare recommends beginning with the Alluvi Retatrutide 20mg Pen for the first 8 weeks of your initiation and first titration protocol. This provides complete baseline receptor response data and tolerance documentation before transitioning to the Alluvi Retatrutide 40mg Pen for titration-II and maintenance phases from week 9 onwards. Starting with the 40mg pen and skipping the initiation phase sacrifices critical baseline data.
Yes — in a research context. Many UK researchers use the Alluvi Retatrutide 20mg Pen and Synedica Tirzepatide in parallel or sequential comparative studies to investigate the incremental effects of adding glucagon receptor agonism (present in retatrutide, absent in tirzepatide) to the GLP-1/GIP dual pathway. Both are available from Alluvi Healthcare with COAs and Synedica certification.
Store refrigerated at 2–8°C. Do not freeze under any circumstances. Protect from direct light. Unopened shelf life is up to 24 months from manufacture date. Once opened, the pen may be kept at room temperature (below 30°C) for up to 28 days, or returned to refrigeration between research sessions.
Retatrutide is available for purchase in the UK as a research compound for laboratory use only. The Alluvi Retatrutide 20mg Pen is not licensed as a medicine under MHRA regulation and is not available as a prescription product. It is supplied strictly as a research compound. Not for human consumption. Purchasers are responsible for compliance with all applicable institutional and regulatory requirements. For regulatory context see: mhra.gov.uk.
Explore More from Alluvi Healthcare
🏠


Reviews
There are no reviews yet.